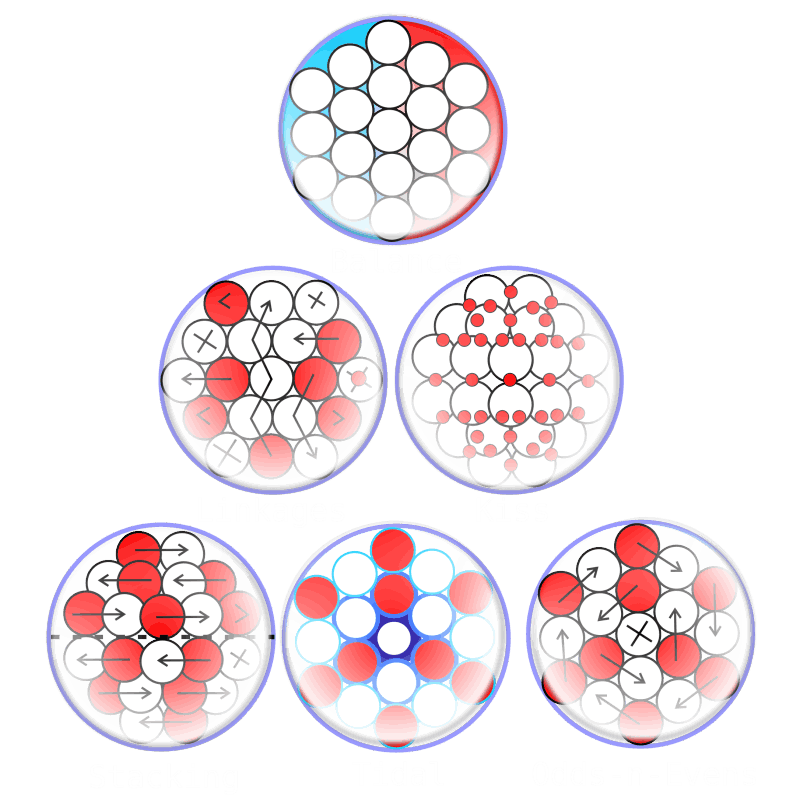
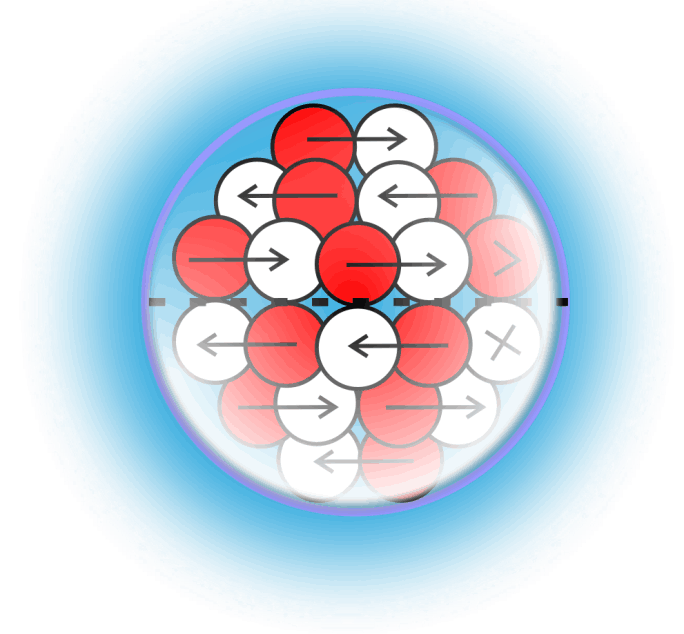
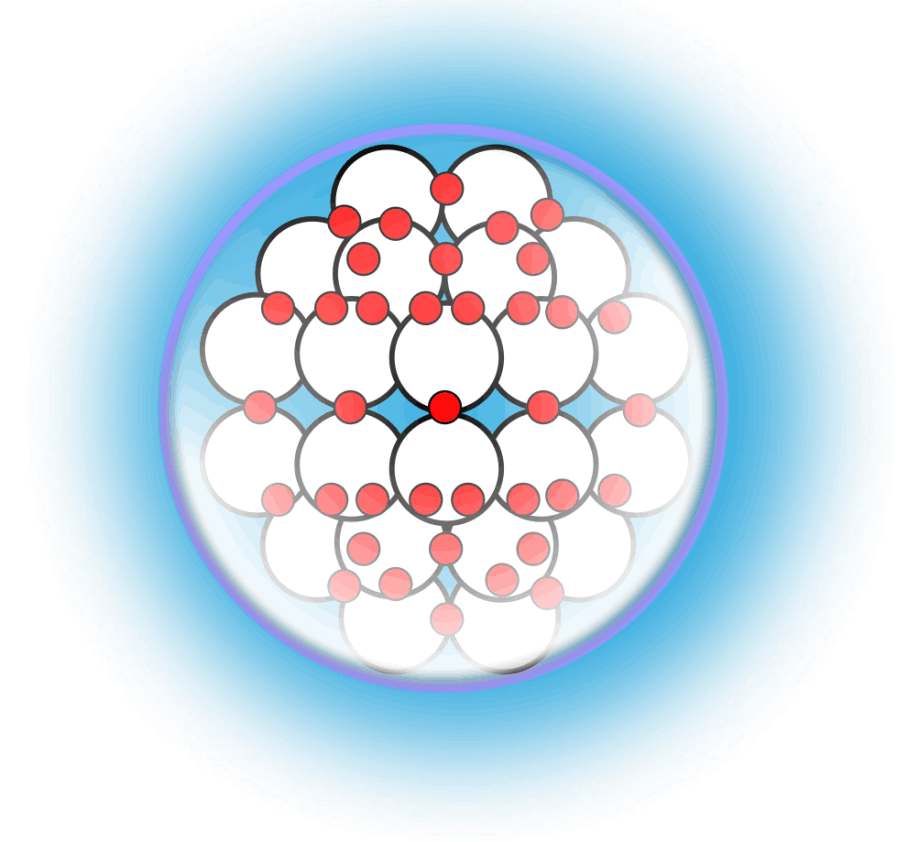
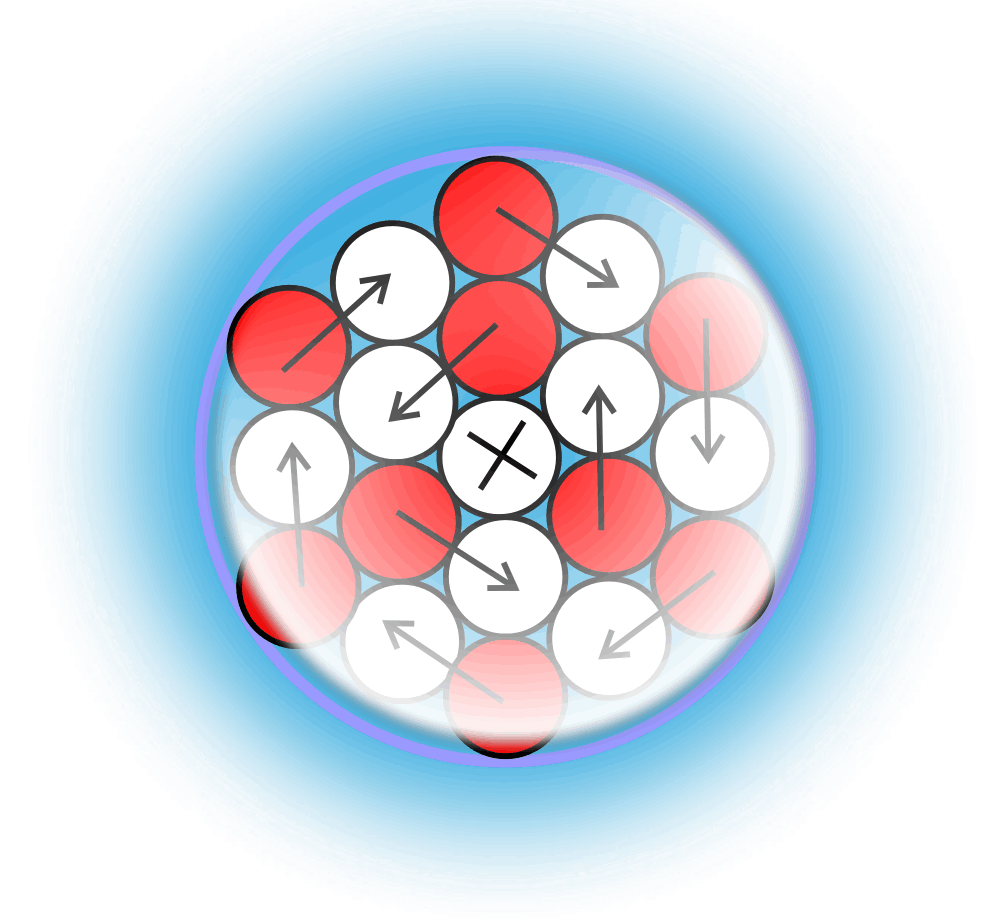
Liquid Gravity
Atomic Structure
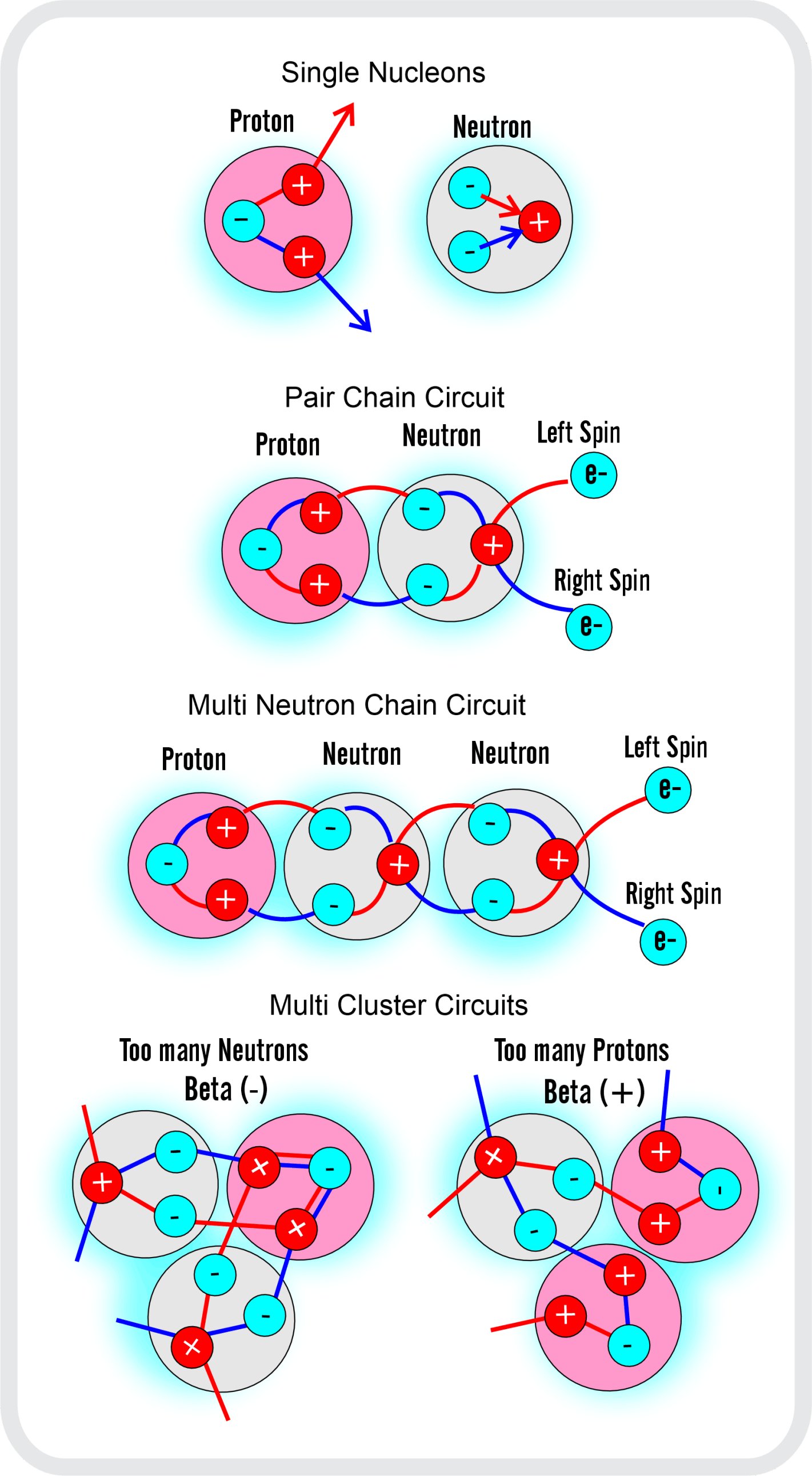
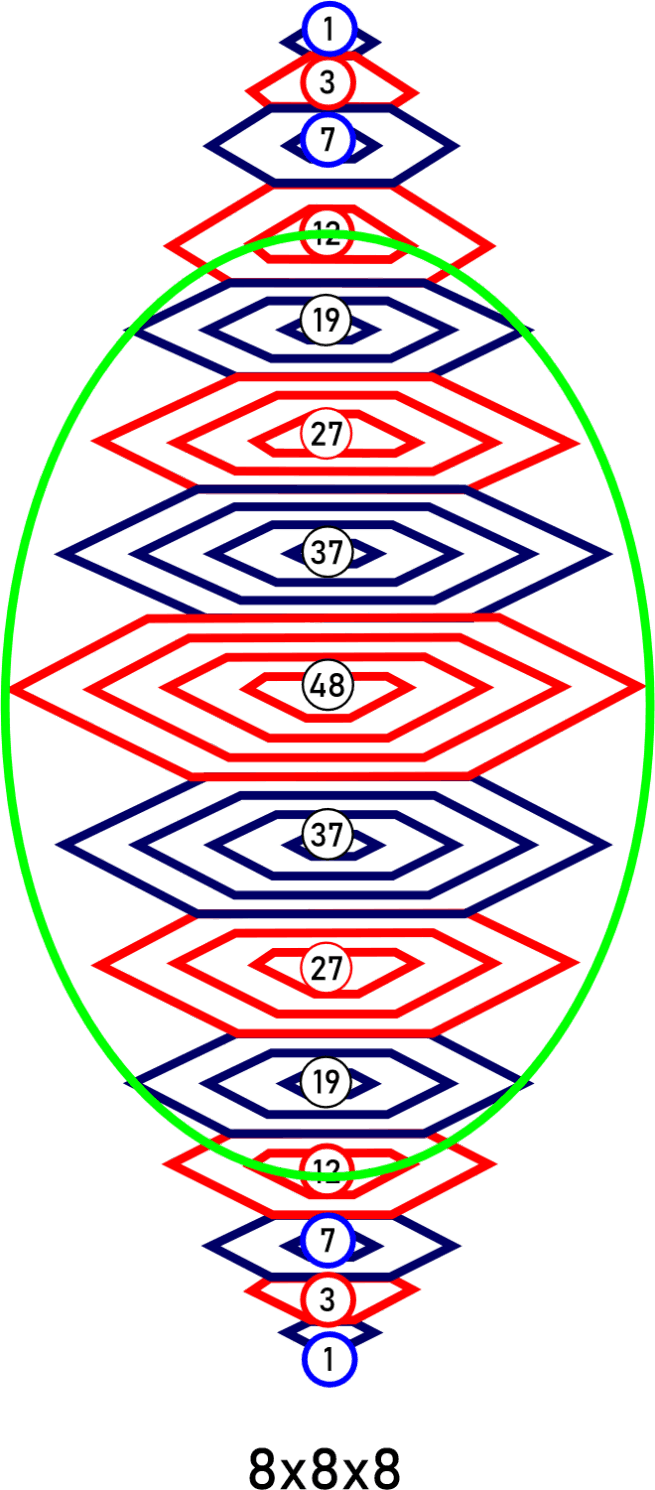
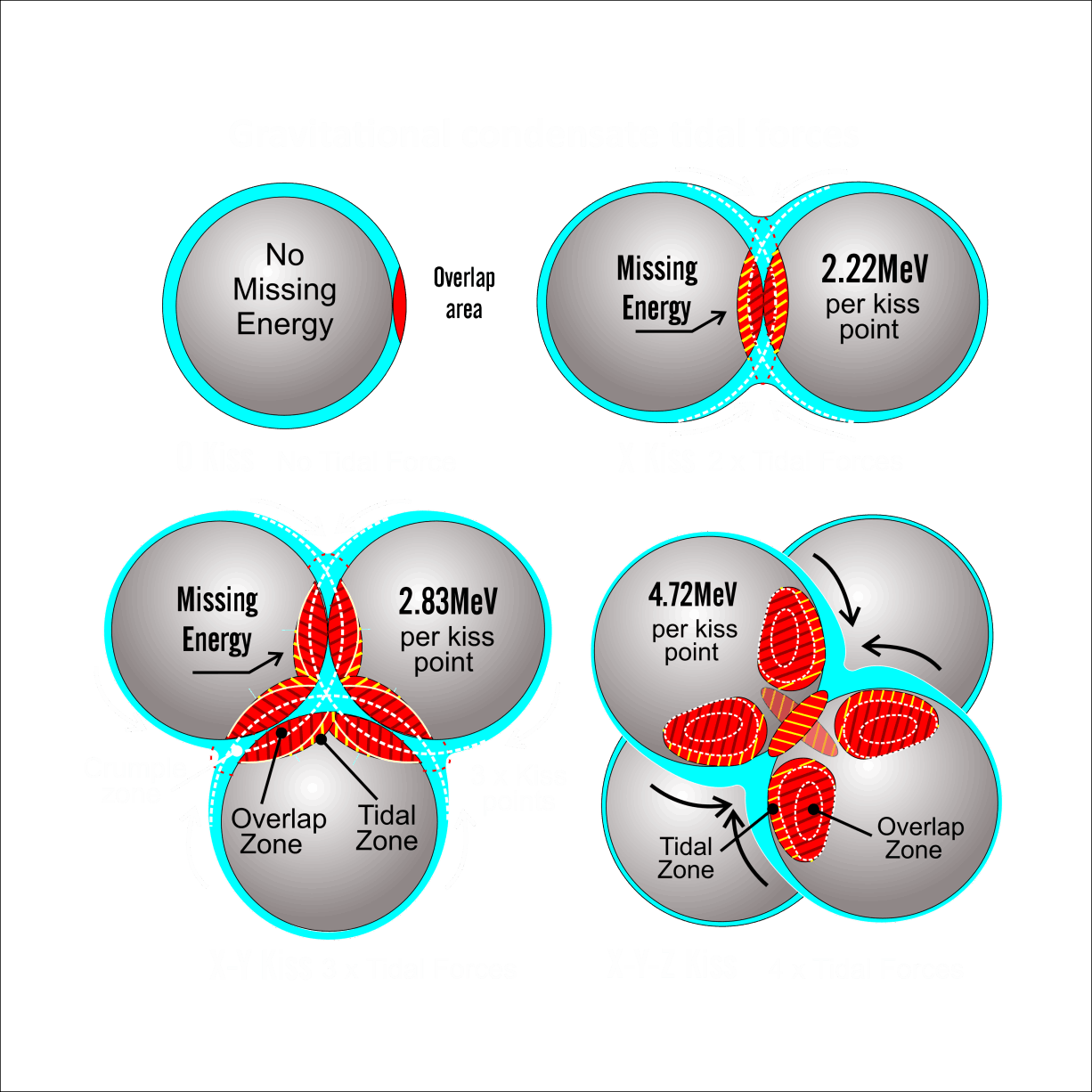
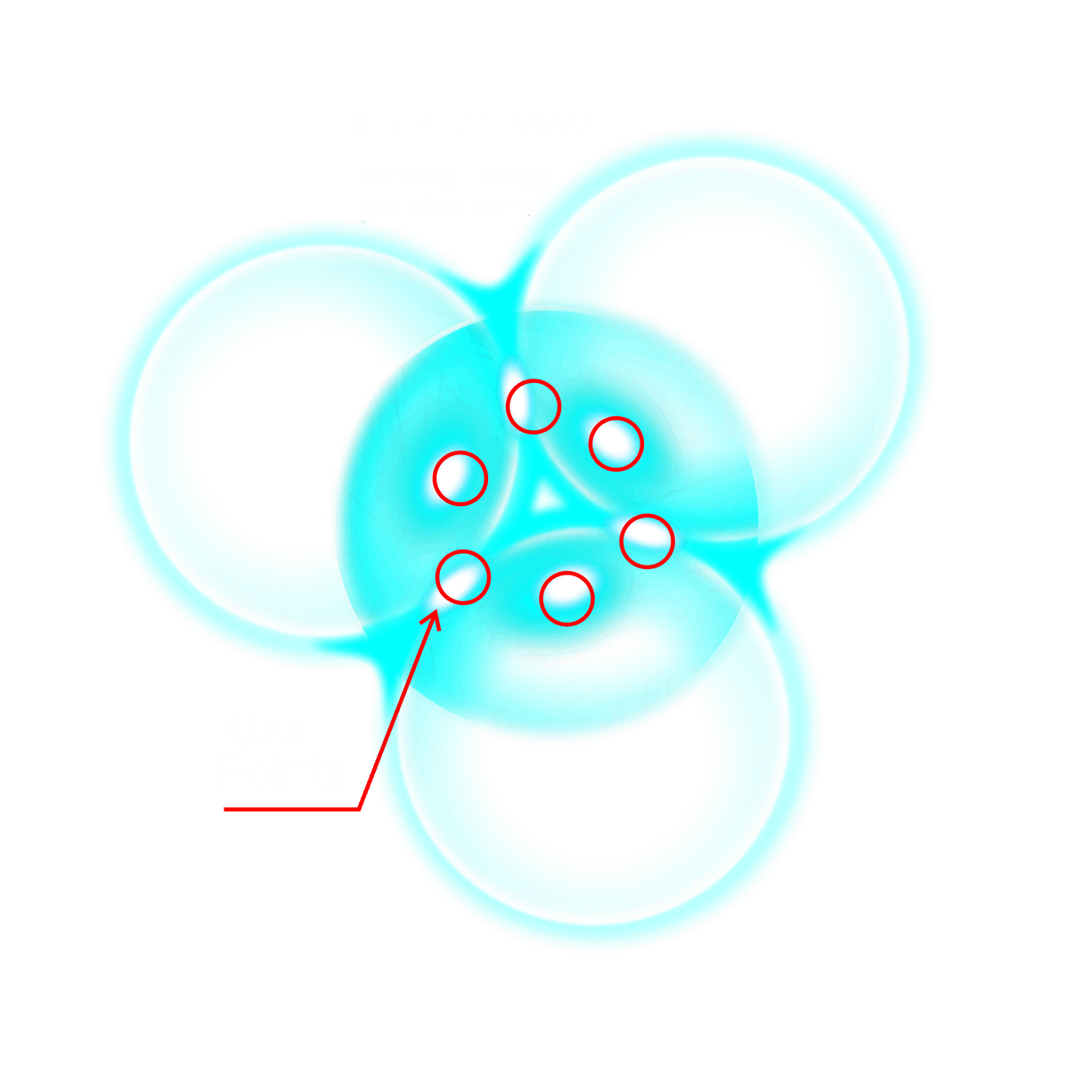
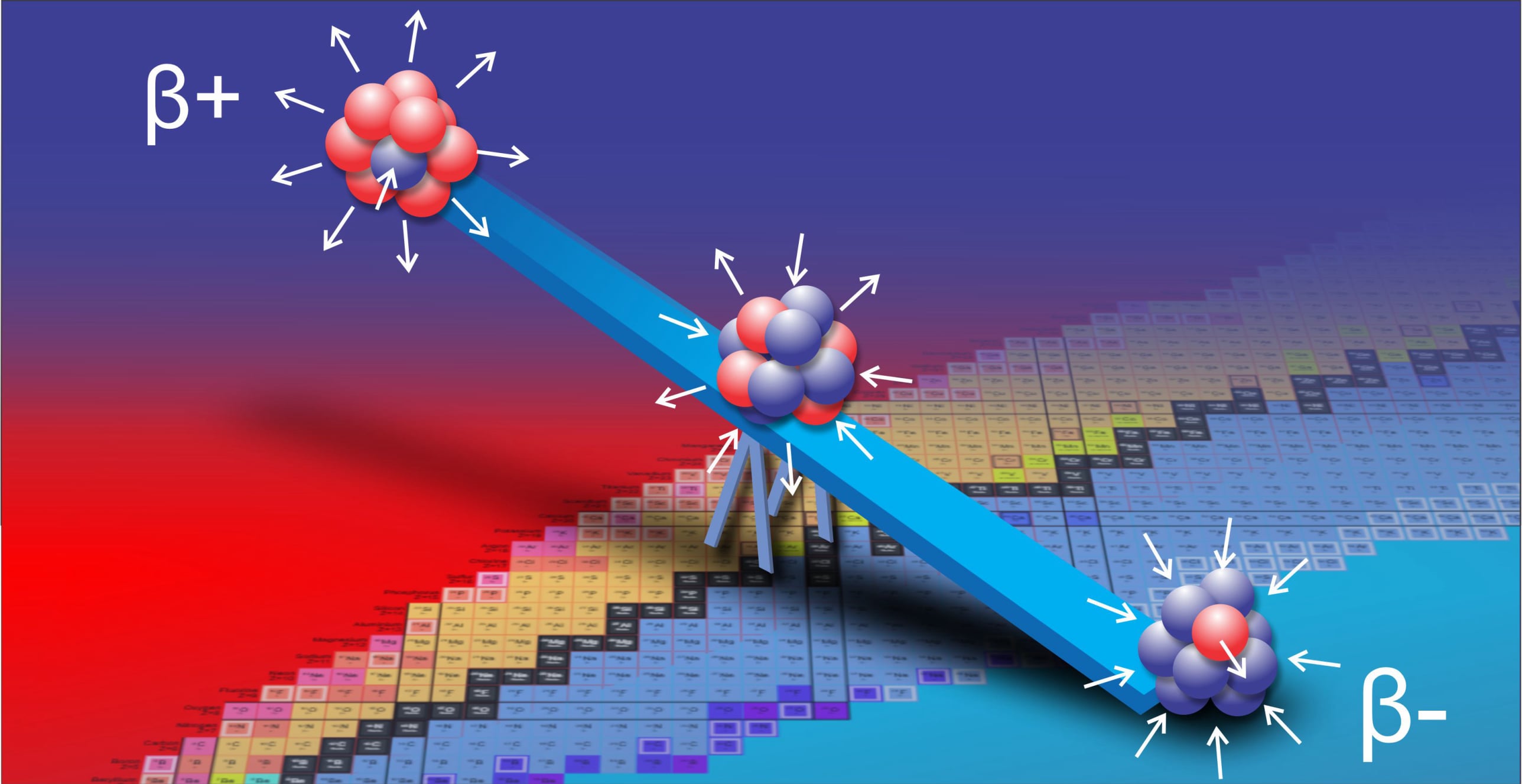
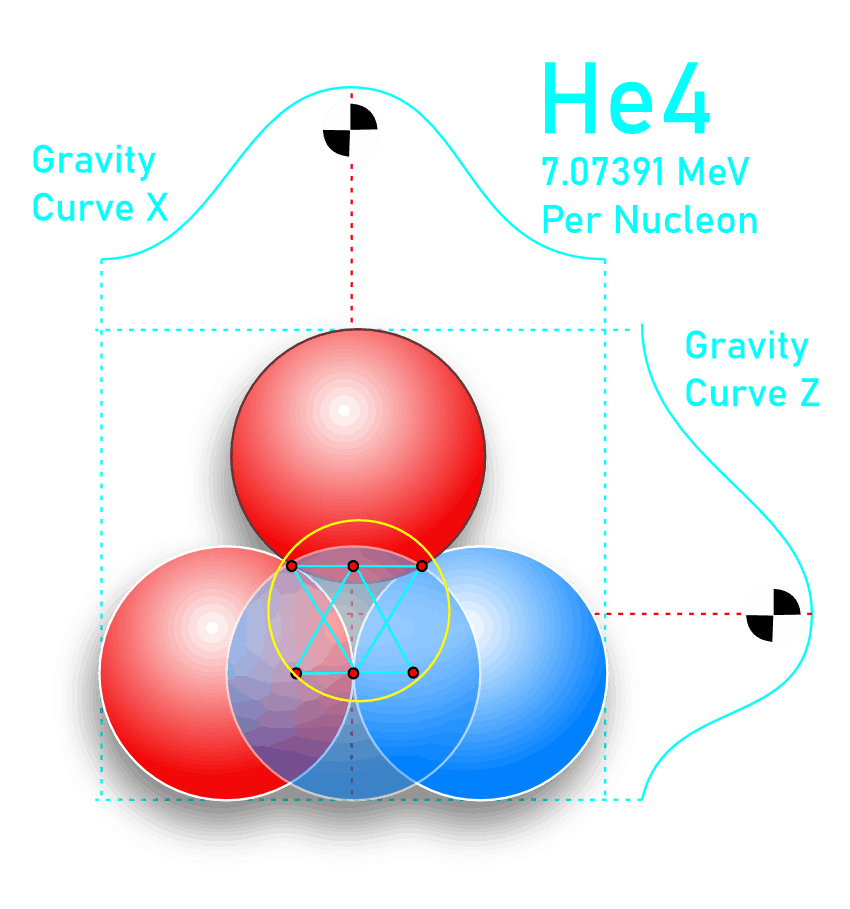
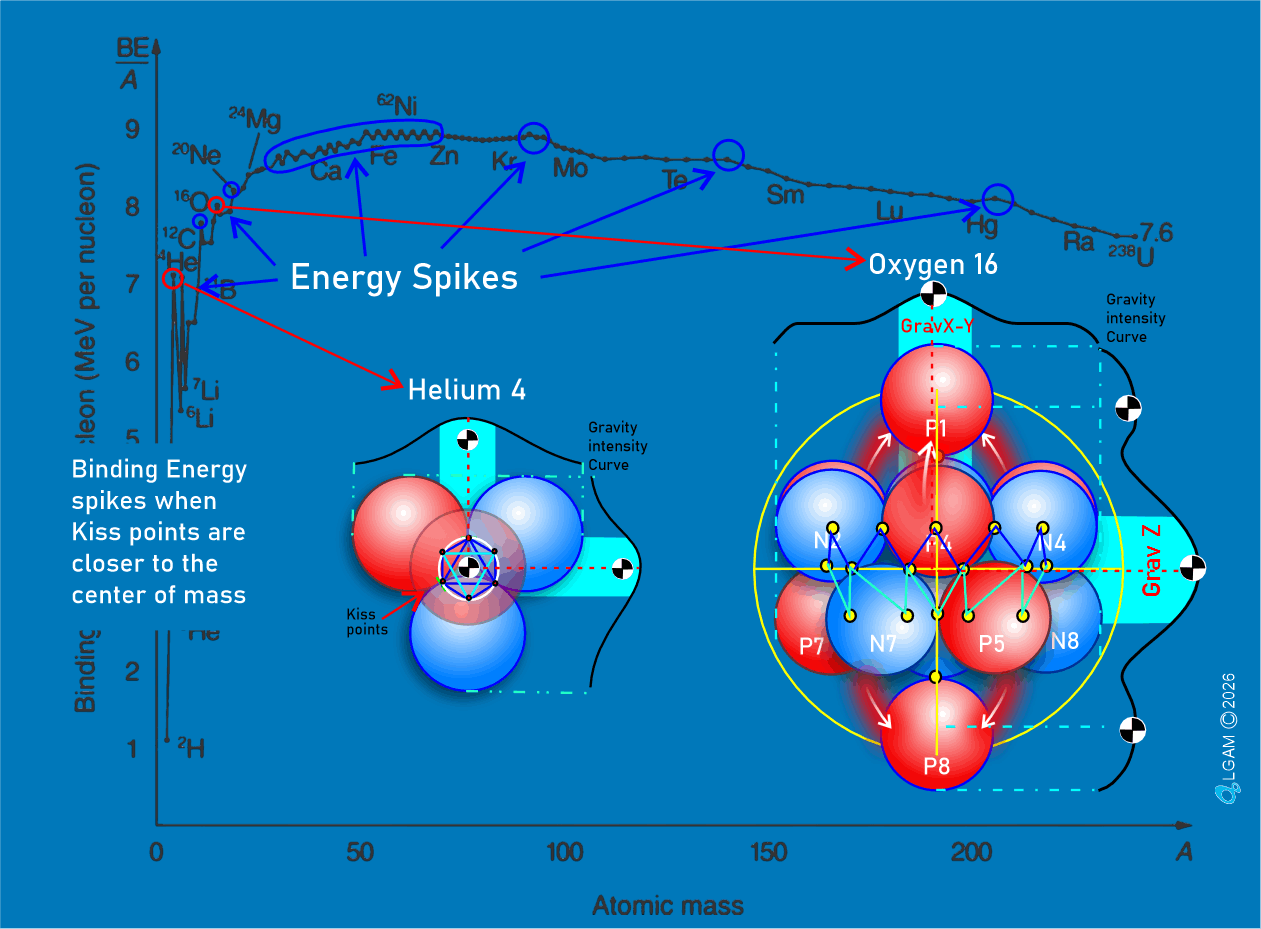
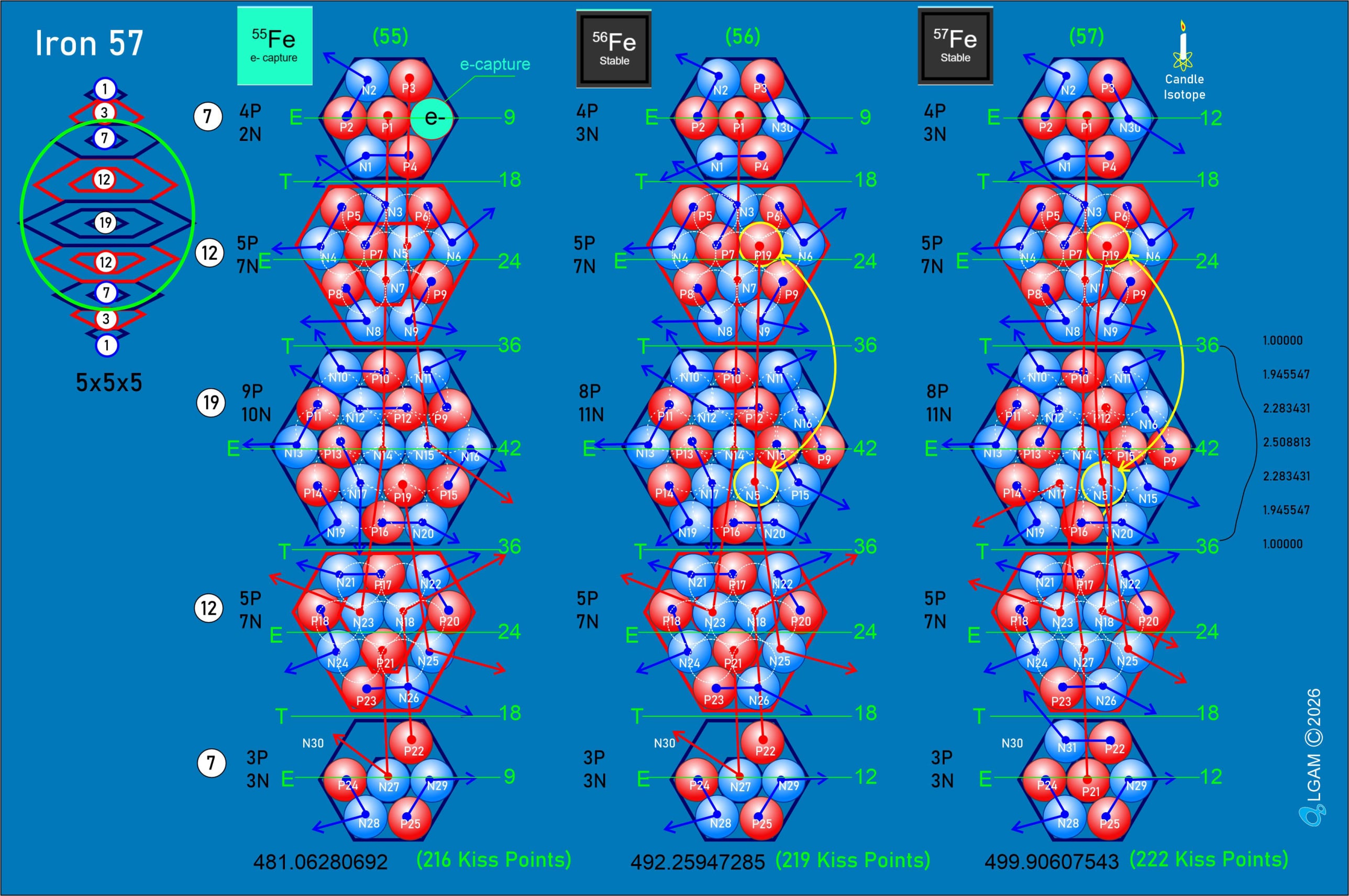
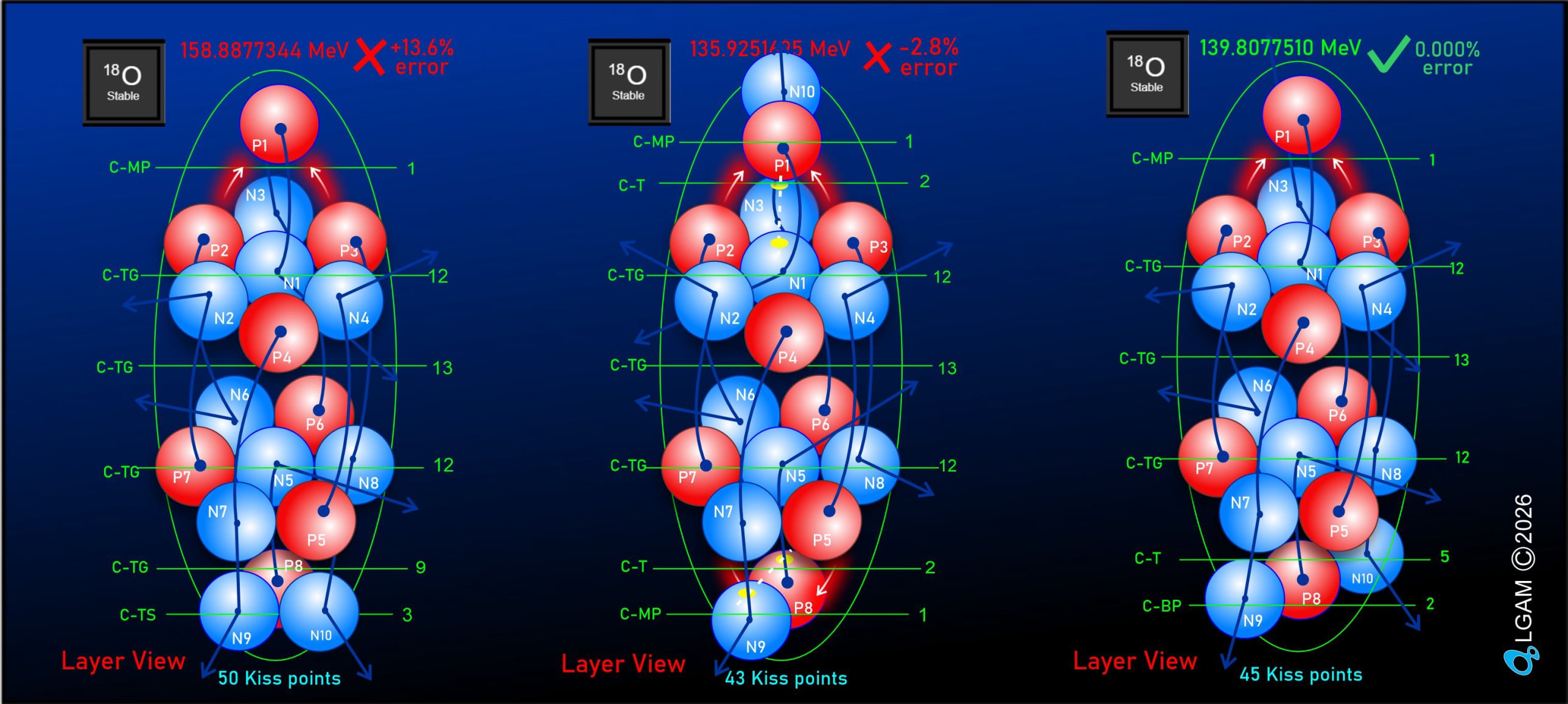
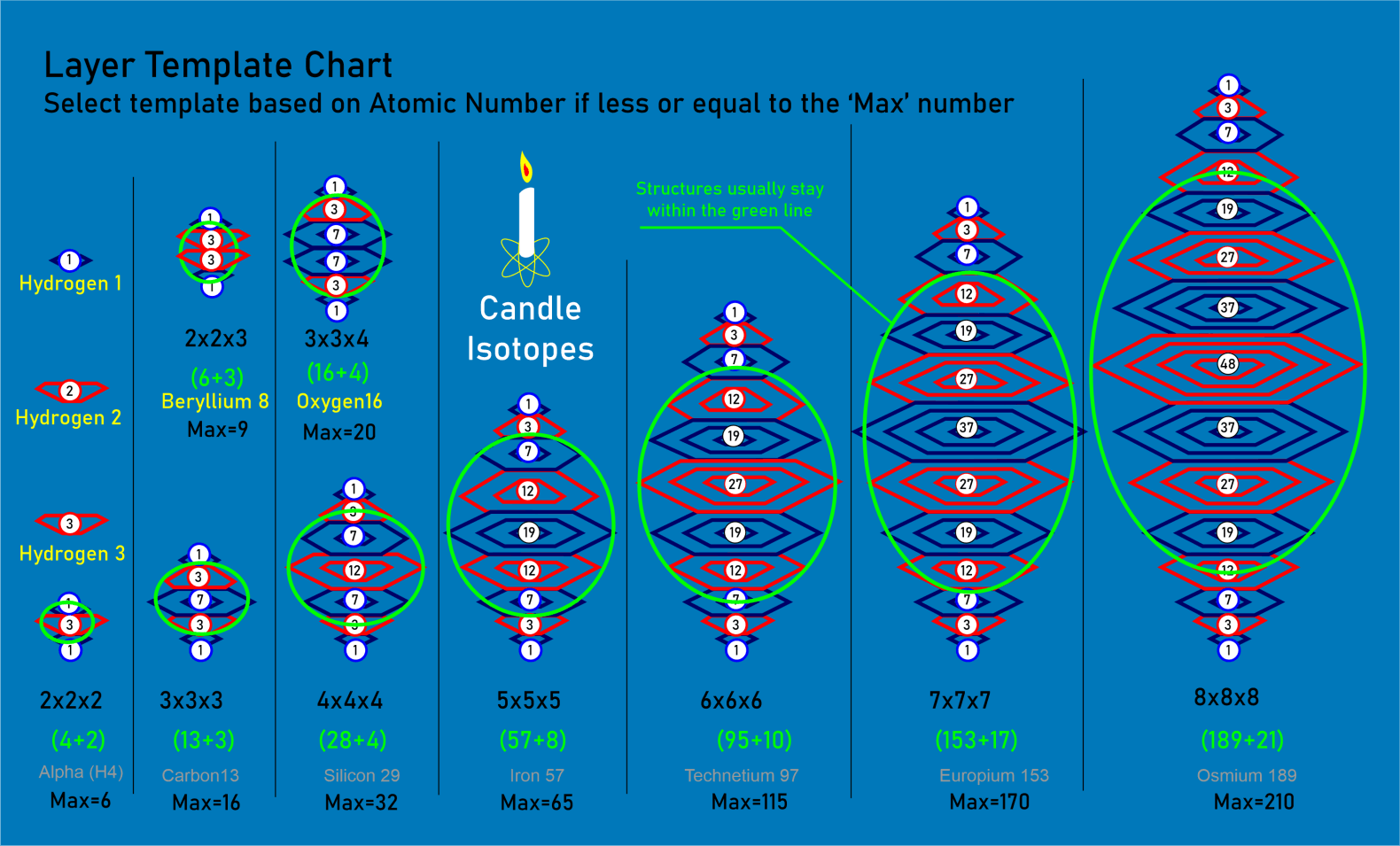
For decades progress on atomic structure has been limited by the prevailing view that nuclei are loosely bound, vibrating collections of nucleons randomly exchanging gluons and quarks—a liquid‑drop picture that yields useful semi‑empirical fits but few reliable predictions. The LGA model instead identifies explicit local mechanisms that power atomic structure and uses them to make more accurate, testable predictions.