Carbon
LGA Model
6 key properties govern the structure and binding energy of a nucleus. Understanding these will help science explore the hidden behavior of atoms and lead to new discoveries.
6 key properties govern the structure and binding energy of a nucleus. Understanding these will help science explore the hidden behavior of atoms and lead to new discoveries.

Carbon Isotopes 11–14 have been modeled from binding data by applying the principles of the Liquid Gravity Atomic (LGA) model (see the step-by-step methodology here).
By utilizing empirical measurement data to guide the structural construction of each isotope, this method reveals their precise geometric configurations. Evaluating these structures side-by-side ensures consistency across the entire isotope family. The resulting models provide a highly plausible visualization of each isotope's structure, offering structural explanations for their unique physical properties.
Results
As summarized below, the results speak for themselves—demonstrating a staggering increase in predictive accuracy compared to conventional frameworks.
Conclusion
The LGA model improves structural binding accuracy for Carbon 11–14 by 5.5 orders of magnitude compared to the traditional Liquid Drop Model (LDM).
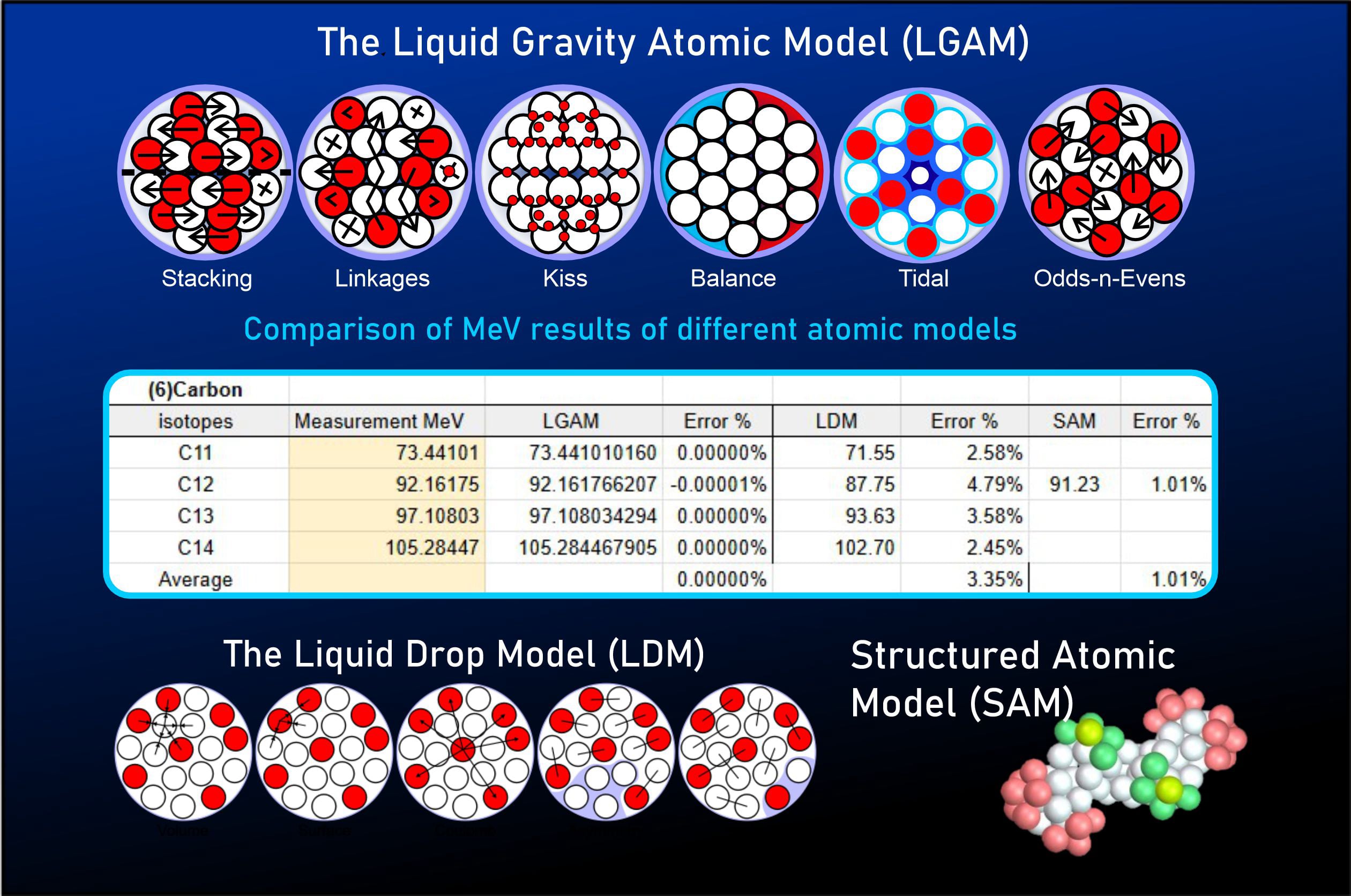
Mainstream science, primarily utilizing the Liquid Drop Model, does not attempt to model an atom’s nucleus with structural precision. This is based on the premise that the nucleus is comprised of a collection of loosely packed nucleons that randomly pulsate within a confined space. Conversely, the Structured Atomic Model (SAM) does attempt to define a specific structure based on its own set of rules; however, it relies on an approximate method for calculating binding energy that fails to accurately reproduce observed data. While Quantum Chromodynamics (QCD) should theoretically be able to calculate binding energies, physicists claim the calculations are too vast and complex for existing technology (though it is possible the underlying premise itself is flawed).
The Liquid Gravity Atomic Model utilizes existing data to build a profile of interactions between nucleons. These rules can be scaled across the entire spectrum of elements and their isotopes. The examples below analyze Carbon-11 through Carbon-14, identifying their unique structures by reverse-engineering observed binding data.
Notable Features:
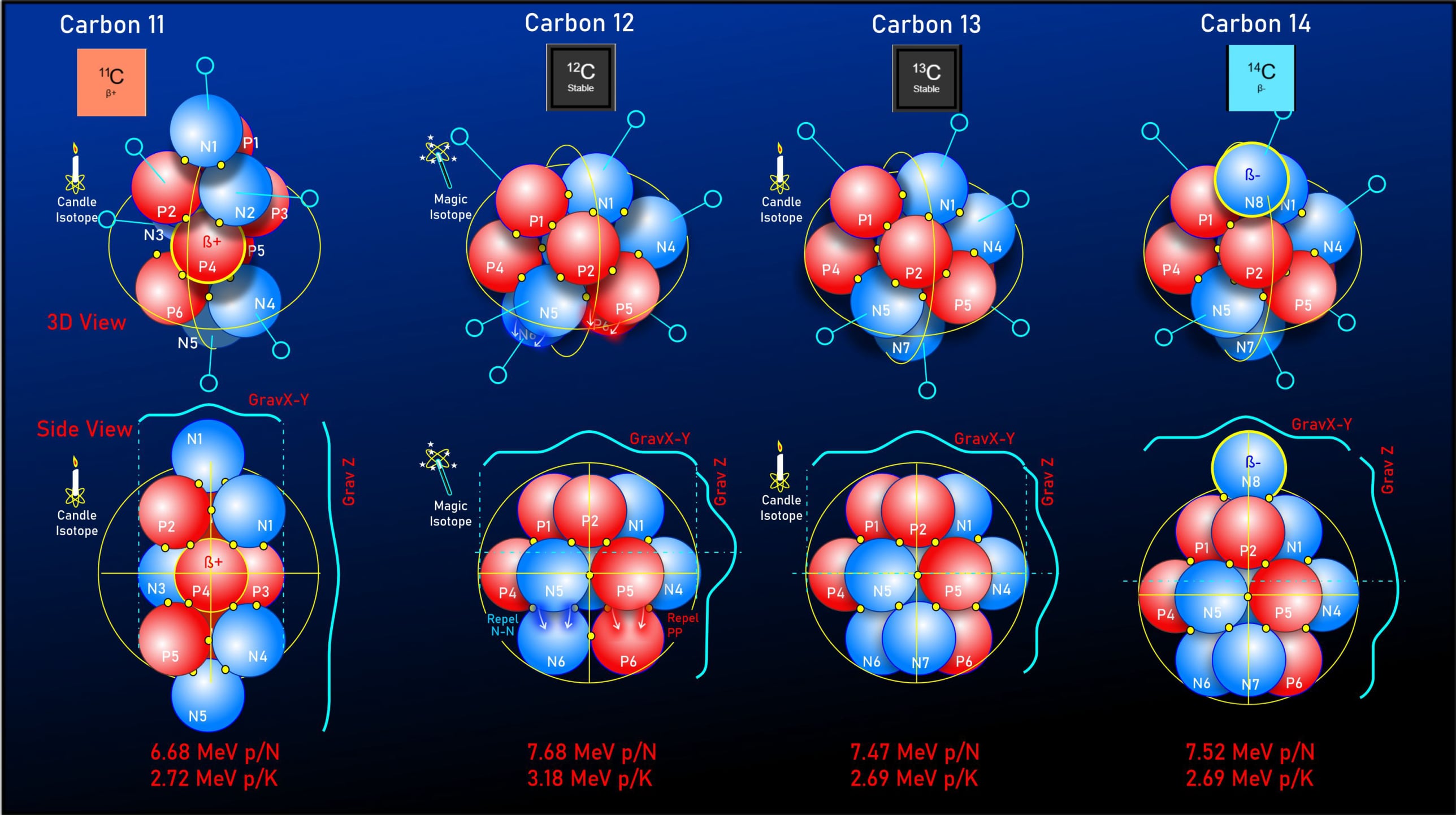
Carbon-11
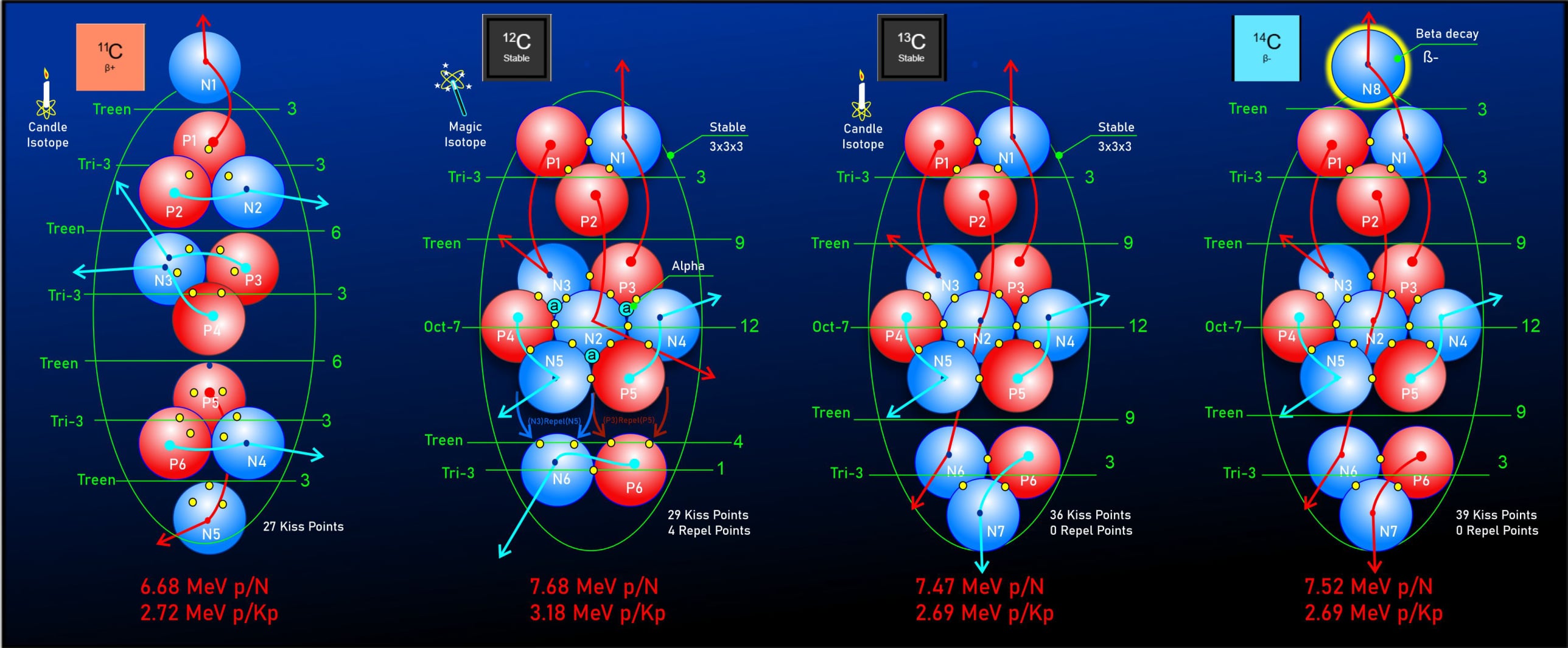
Carbon-11 is built on three tri-structured layers, capped by single nucleons at both the top and bottom. This elongated 3*5 formation is unusual, as it sits outside the standard "stability rule" which dictates that the layer count should equal the width count. Despite this irregularity, this configuration provides a more accurate binding energy result than alternative tested models.
Detailed analysis of this structure reveals a specific pairing of protons to a single nucleon at each level, with the central layer forming a Helium-3 structure that shares a common circuit. Compared to other carbon isotopes, Carbon-11 exhibits the lowest MeV per nucleon. Within this framework, this low energy profile is attributed to a gravitational geometry that is stretched long and thin, which results in lower tidal margins and subsequently reduced binding energy.
Furthermore, Carbon-11 operates as a "Candle Isotope." Its symmetrical geometry provides a perfectly uniform distribution of kiss points; these can be divided directly by the total gravitational MeV to precisely calculate individual kiss point values.
Carbon-12
Carbon-12 is often noted for its exceptional stability compared to its neighboring isotopes. Ironically, neither 6 nor 12 are standard nuclear "magic numbers"; however, mainstream science attributes this stability to Carbon-12 being structurally related to three alpha particles, which themselves are composed of magic numbers.
Conversely, the LGA model explains this increase in binding energy through a mechanism called "cresting"—a structural feature characteristic of highly stable isotopes. This formation occurs on the top or bottom structural layers when single protons and neutrons are repelled by nucleons on the adjacent layer, driven by the electromagnetic repulsion of like charges. The result is a weaker gravitational "crest" connection rather than a stronger "triple nest" connection. This structural shift, in turn, allows the main cluster of nucleons to consolidate their gravitational force around the upper "kiss points," deepening the tidal effect and increasing overall binding energy.
While three alpha particles may initially collide to form Carbon-12, the resulting ground-state structure and binding energies are significantly different, yielding over 7MeV of additional binding energy. Therefore, rather than attributing this unique stability strictly to traditional magic numbers, the LGA model demonstrates a direct correlation between specific structural formations and gravitational consolidation as the primary drivers of increased atomic binding energy.
Carbon-13
Carbon-13 features a highly uniform nucleus, providing another "Candle Isotope" that allows for the precise calculation of individual kiss point values through its symmetric formation. While its core architecture is structurally similar to Carbon-12, Carbon-13 exhibits a lower binding energy. Within this framework, this decrease is attributed to the manner in which an additional neutron stabilizes the bottom layer into a nesting configuration. Consequently, the gravitational profile becomes evenly distributed in both directions; this shifts the central kiss points further away from the center of mass, reducing tidal margins and lowering the overall binding energy.
Carbon-14
Carbon-14 maintains nearly the same average kiss point MeV as Carbon-13, showing only a minor variation introduced by an extra neutron capping the top layer. This addition slightly elongates the gravitational profile around the upper kiss points. While this structural shift pushes Carbon-14 into an "unbalanced" state, the underlying stability of the arrangement ensures it remains a exceptionally slow beta-decayer. This stable yet unbalanced configuration gives Carbon-14 its characteristic half-life of roughly 5,700 years, making it globally essential for radiocarbon dating.
Mainstream science suggests that protons carry a positive charge while neutrons remain neutral, implying that the strong nuclear force is the sole mechanism holding the nucleus together against the electrostatic repulsion of protons.
The Liquid Gravity Atomic Model (LGAM) introduces a paradigm shift. We propose a new way to understand electrical forces, viewing protons and neutrons as collaborative components that form integrated nuclear circuits. While the broader evidence for this is explored in our Atomic Structure section, this page focuses specifically on the circuits discovered within the four carbon isotopes.
The Mechanics of Nuclear Circuits
In our models, nuclear architecture is not random; it follows specific geometric and physical rules:
Proton Distribution:
Because protons naturally repel one another, they distribute themselves as widely as the cluster geometry allows.
Gravitational Compression:
Countering this repulsion is the force of gravity, which works to pull nucleons into a dense, cohesive clump.
Quark Orientation:
Inside each nucleon, quarks with varying degrees of positive and negative charge create internal polarity. These charges orient the nucleons, allowing opposite quark charges to attract and establish a "circuit path" ready to receive the charge pulse from the protons.
Carbon Isotope Architecture
We observe consistent patterns of proton and neutron distribution across adjacent isotopes because they are already in optimal positions. However, carbon reveals a significant structural step-change:
Carbon-11: Features a compact, 3-nucleon tri-structure core.
Carbon-12: Introduces a major shift to an octagon geometry with a 7-nucleon core.
This structural evolution in Carbon-12 changes the entire configuration, and the subsequent carbon isotopes follow this modified 7-nucleon core structure.
Mainstream science uses a combination of the Liquid Drop Model and the Shell Model to predict the binding energy of isotopes; however, these models typically produce average results rather than identifying the binding data of individual nucleons. Using existing theories, attempts to calculate individual nucleon binding data remain computationally over-complex.
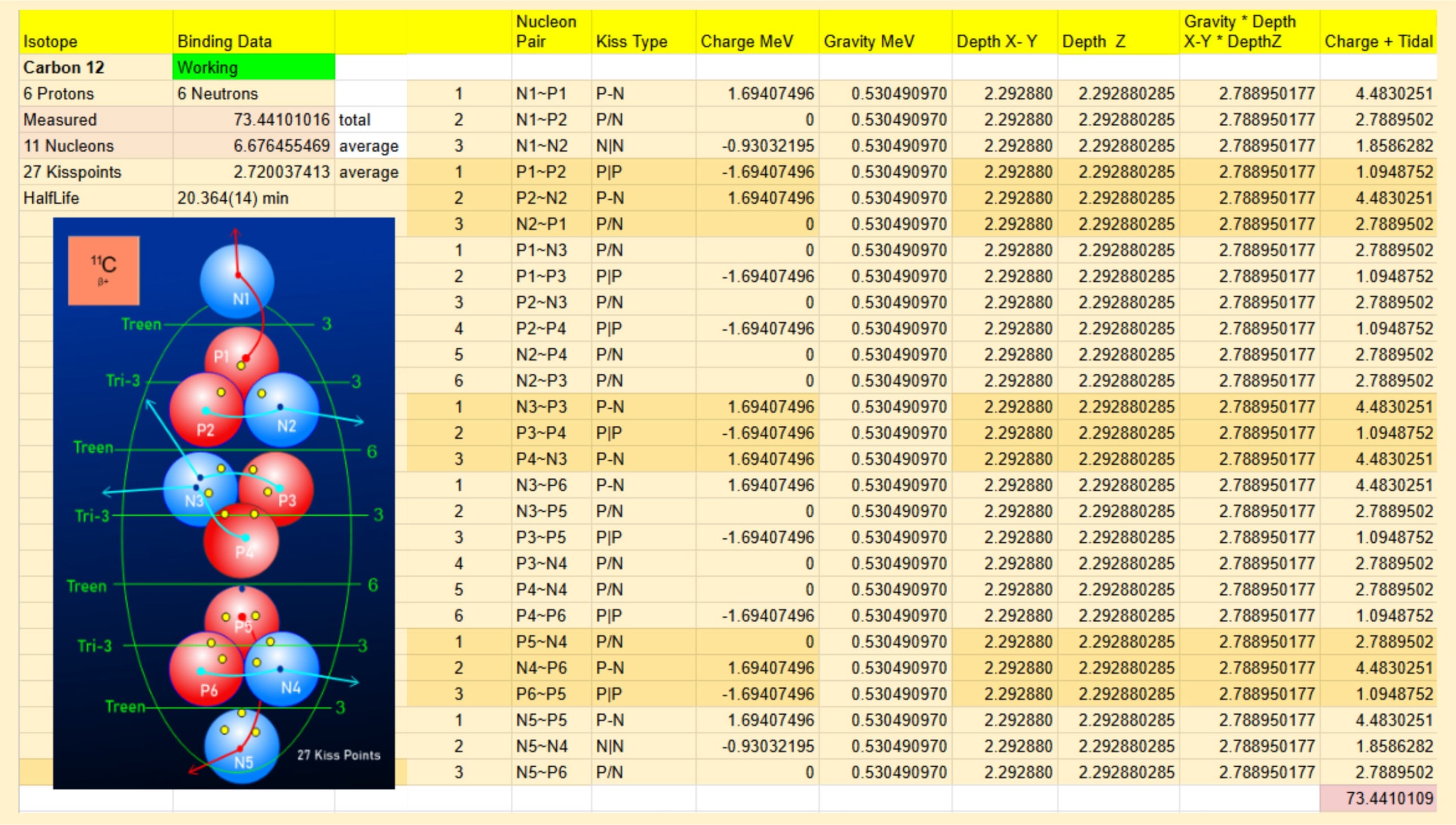
In contrast, the Liquid Gravity Atomic (LGA) Model utilizes a structural approach governed by the rules of electromagnetic attraction/repulsion and gravitational forces. Once the model is constructed, the "Kiss Points"—where nucleons physically contact one another—are cataloged and their relationships identified. By mapping these locations, the value of each individual "kiss point" is calculated and then summed to compare against empirical measurements. This method produces a highly accurate map of all contact points, providing a structural lattice of known values that offers insight into the specific strengths or weaknesses of a nuclear structure.
Carbon-12 serves as a "candle isotope," providing a baseline measurement due to its uniform and symmetrical structure. These values can then be applied to other isotopes that share similar regions but lack symmetry. Just as astronomers use Type 1a supernovae as "standard candles" to measure distances in space, the LGA Model identifies candle isotopes to establish the foundational measurements for its calculations.
Term Definition: Candle Isotope
An LGA Model term used to classify specific nucleon structures (such as Carbon-11 and Carbon-13) that possess an exceptionally uniform and symmetrical geometry. Because of this structural uniformity, a Candle Isotope serves as a reliable baseline for calculating individual nucleon binding forces.
The term is adapted from the "Standard Candles" of astronomy—cosmic objects with a known luminosity used as fixed benchmarks to measure vast interstellar distances. Similarly, in the LGA framework, a Candle Isotope provides a predictable distribution of "kiss points" that can be divided directly by the total gravitational MeV to accurately determine individual kiss point values.